* The diazo compound may follow either one of the two pathways depending on the reaction conditions. :max_bytes(150000):strip_icc()/Bamford-Stevens-Reaction-58b5e60a5f9b58604604ae28.png)
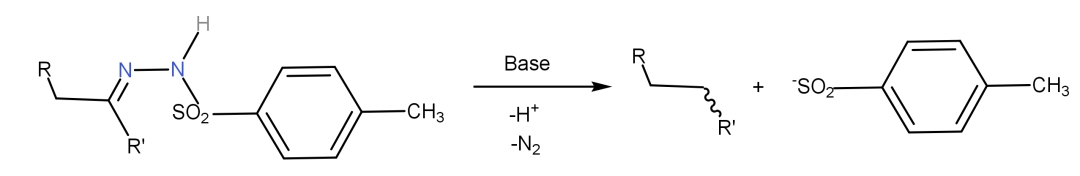
Initially, the reaction of tosyl hydrazone with a strong base leads to a diazo compound, which can be isolated in some cases. This reaction employs bases such as alkyllithiums and Grignard reagents. * Whereas in Shapiro reaction, the less substituted alkene is formed as the kinetic product. * In case of Bamford-Stevens reaction, the more substituted alkene is formed as the thermodynamic product. * Both the Bamford-Stevens reaction and the Shapiro reaction afford alkenes from tosyl hydrazones. * The reaction may be performed either in protic solvents like glycols or in aprotic solvents like ethylene glycol dimethyl ether. * In the Bamford-Stevens reaction, the tosyl hydrazones (p-Toluenesulfonyl hydrazones) of aliphatic aldehydes or ketones furnish more substituted alkenes when treated with strong bases like NaOMe, NaH, LiH, NaNH 2 etc.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
